Hemogen announced the publication of clinical results for its lead gene therapy candidate, hemo-cel (HGI-001 injection), in the peer-reviewed journal Molecular Therapy. The study reported long-term outcomes in patients with transfusion-dependent β-thalassemia (TDT), demonstrating sustained transfusion independence and durable therapeutic effect exceeding five years of follow-up (as of February 2026).
These data establish clinical proof-of-concept for hemo-cel as a potentially curative autologous hematopoietic stem cell gene therapy and support its continued global clinical and commercial development.

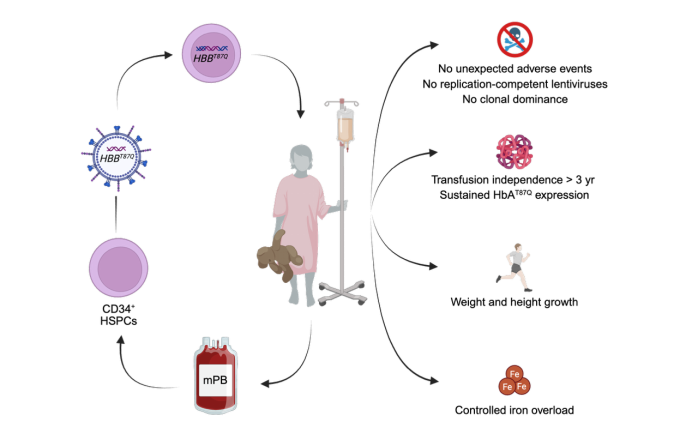
Adapted from Liu et al., Molecular Therapy, 2026
Clinically Validated Durable Efficacy Demonstrates Competitive Therapeutic Profile
Hemo-cel is an autologous hematopoietic stem cell gene therapy utilizing lentiviral vector–mediated delivery of a functional β-globin gene into patient-derived stem cells, enabling long-term restoration of physiological hemoglobin production.
Key clinical outcomes include:
· Rapid hematopoietic recovery, with neutrophil engraftment achieved within 16-22 days and platelet engraftment within 12-19 days
· Sustained transfusion independence for 38–61 months, with maximum follow-up exceeding five years (as of February 2026)
· Normalization of weight and height trajectories
· Effective control of iron overload following discontinuation of iron chelation therapy
The durability of response observed across extended follow-up provides important validation of long-term therapeutic stability, a critical requirement for regulatory approval and commercial adoption of gene therapies.
These results position hemo-cel as a differentiated therapeutic candidate addressing the significant unmet medical need in transfusion-dependent β-thalassemia.
Favorable Long-Term Safety Profile
Comprehensive long-term safety monitoring demonstrated:
· No detection of replication-competent lentivirus
· No evidence of clonal dominance or insertional oncogenesis
· No serious adverse events attributable to vector integration
Integration site analysis confirmed polyclonal hematopoietic reconstitution, supporting the long-term safety and stability of gene-modified stem cells.
These findings support a favorable benefit-risk profile and strengthen regulatory readiness across multiple global jurisdictions.
Advancing Global Clinical Development and Scalable Deployment Through SPARK
In parallel with clinical advancement, Hemogen has developed SPARK, a proprietary end-to-end global deployment model designed to enable compliant, scalable, and localized implementation of advanced gene therapies across international markets.
The SPARK framework integrates five core pillars:
S — Service & Support
P — Platform
A — All-in-One Kits
R — Regulatory
K — Knowledge & Innovation
These components form a structured solution that supports manufacturing transfer, regulatory readiness, clinical implementation, and long-term capability building in emerging and high-prevalence regions.
Through SPARK, Hemogen enables efficient localization of gene therapy manufacturing and clinical implementation while ensuring product consistency, quality, and regulatory compliance across diverse healthcare systems.
Hemogen is advancing hemo-cel through parallel global pathways, including:
· IND approval obtained in China, with registered clinical trials actively progressing
· Clinical trial authorization from Thailand FDA (TFDA), with overseas manufacturing infrastructure established
· Clinical application under technical review by Saudi FDA (SFDA)
· Strategic expansion across Middle Eastern markets, including Jordan and Bahrain
Fully Integrated Platform Supports Scalable Manufacturing and Pipeline Expansion
Hemogen has established a vertically integrated gene therapy platform spanning vector development, cell processing, analytics, and long-term safety monitoring, including:
· Proprietary lentiviral vector design and gene transfer technologies
· Automated cell processing and manufacturing system (Celaxi-23)
· Integration site analysis and long-term molecular monitoring platform
· CRISPR/Cas gene editing capabilities supporting next-generation pipeline development
This integrated infrastructure supports:
· Scalable manufacturing readiness
· Robust CMC and quality control systems
· Long-term safety validation required for regulatory approval
· Platform expansion into additional indications across multiple disease areas
Hemogen holds 17 patents and has received multiple national and regional innovation recognitions, reflecting its technological leadership and intellectual property foundation.
Positioned for Strategic Partnerships and Global Commercial Expansion
With validated clinical efficacy, favorable long-term safety profile, and established manufacturing and analytical infrastructure, hemo-cel represents a strategic opportunity for global development and commercialization partnerships.
Importantly, Hemogen’s proprietary SPARK integrated deployment model further differentiates its global positioning. By embedding manufacturing platform transfer, standardized production kits, regulatory documentation systems, and structured technical training into a unified framework, SPARK enables scalable and compliant localization of gene therapy capabilities across diverse healthcare systems.
Supported by both a clinically validated product and a structured global implementation model, Hemogen is actively pursuing collaborations to accelerate:
· Late-stage clinical development and regulatory approval
· Global manufacturing scale-up
· Regional commercialization and market access
· Expansion of pipeline programs leveraging its hematopoietic stem cell gene therapy platform
As gene therapy transitions toward broader clinical adoption, Hemogen’s platform, clinical validation, and global development strategy position the company as a strong partner for organizations seeking to expand their presence in transformative genetic medicines.
Publication
Liu et al. Safety and Efficacy of Hemobeglogene Autotemcel (Hemo-cel) Gene Therapy in Five Patients with Transfusion-Dependent β-Thalassemia. Molecular Therapy. Published online 2026.
