Leveraging years of research and translational experience in hematopoietic stem cell gene therapy (HSC-GT), Hemogen was invited to author the review article “Development, Application and Quality Control of Hematopoietic Stem Cell Gene Therapy”. The review comprehensively summarizes key advances in HSC-GT product development and quality control and was published in January 2026 in the Chinese Journal of Cell Biology as part of the special issue on translational cell and gene therapy.
HSC-GT represents a transformative therapeutic modality with the potential to deliver durable, and in some cases lifelong, clinical benefit following a single treatment. In the review, Hemogen outlines the evolution of gene-modification technologies, the current landscape in which lentiviral vectors and CRISPR-based platforms coexist, and the emerging trends toward more precise, efficient, and in vivo gene-editing solutions. While early clinical applications focused primarily on hematologic disorders, the scope of HSC-GT is rapidly expanding to include metabolic and central nervous system diseases. Quality lies at the heart of successful gene therapy. Hemogen highlights a comprehensive quality framework encompassing hematopoietic stem cell characteristics, gene-modification efficacy, cell purity, and safety. Advances in stem cell biology, high-throughput sequencing, and artificial intelligence are accelerating the development of next-generation quality assessment systems.

Figure 1. Schematic of Hematopoietic Stem Cell Gene Therapy
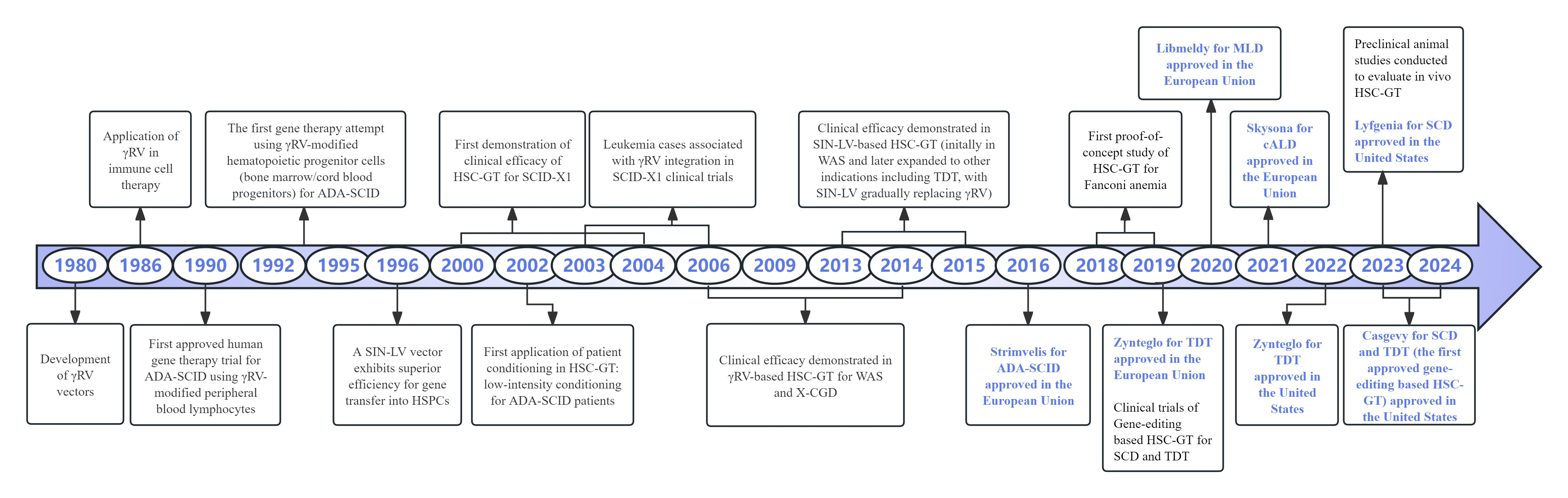
Figure 2. The history of hematopoietic stem cell gene therapy

Figure 3. Framework of quality research and control for HSC-GT products
Hemogen has established both lentiviral and CRISPR/Cas gene-editing platforms and independently developed TarV, an in vivo targeted editing program designed to unlock new therapeutic possibilities. Hemogen’s flagship programs, HGI-001 and HGI-002, have demonstrated favorable safety and efficacy in clinical studies, enabling multiple patients with β- and α-thalassemia to achieve transfusion independence. Meanwhile, additional indications are progressing through preclinical studies. In parallel, Hemogen continues to expand its quality-focused data assets and is leveraging advanced technologies, including single-cell transcriptomics, to build a more precise and comprehensive quality evaluation framework for HSC-GT products.
Looking ahead, Hemogen remains committed to a patient-centered, clinically driven strategy, working to make HSC-GT more precise, more efficient, more accessible, and more reliable for patients worldwide.
To read the full review, please visit: https://www.cjcb.org/cjcb/portal/zl_show.jsp?ID=6311
